TSLL Gain Hunter
TSLL Gain HunterWashington (AP) — The U.S. Food and Drug Administration will hold a meeting this summer to consider relaxing restrictions on more than six types of peptide injections. Peptide injections are a group of unapproved therapies that are widely popular among health influencers, fitness experts, and celebrities.
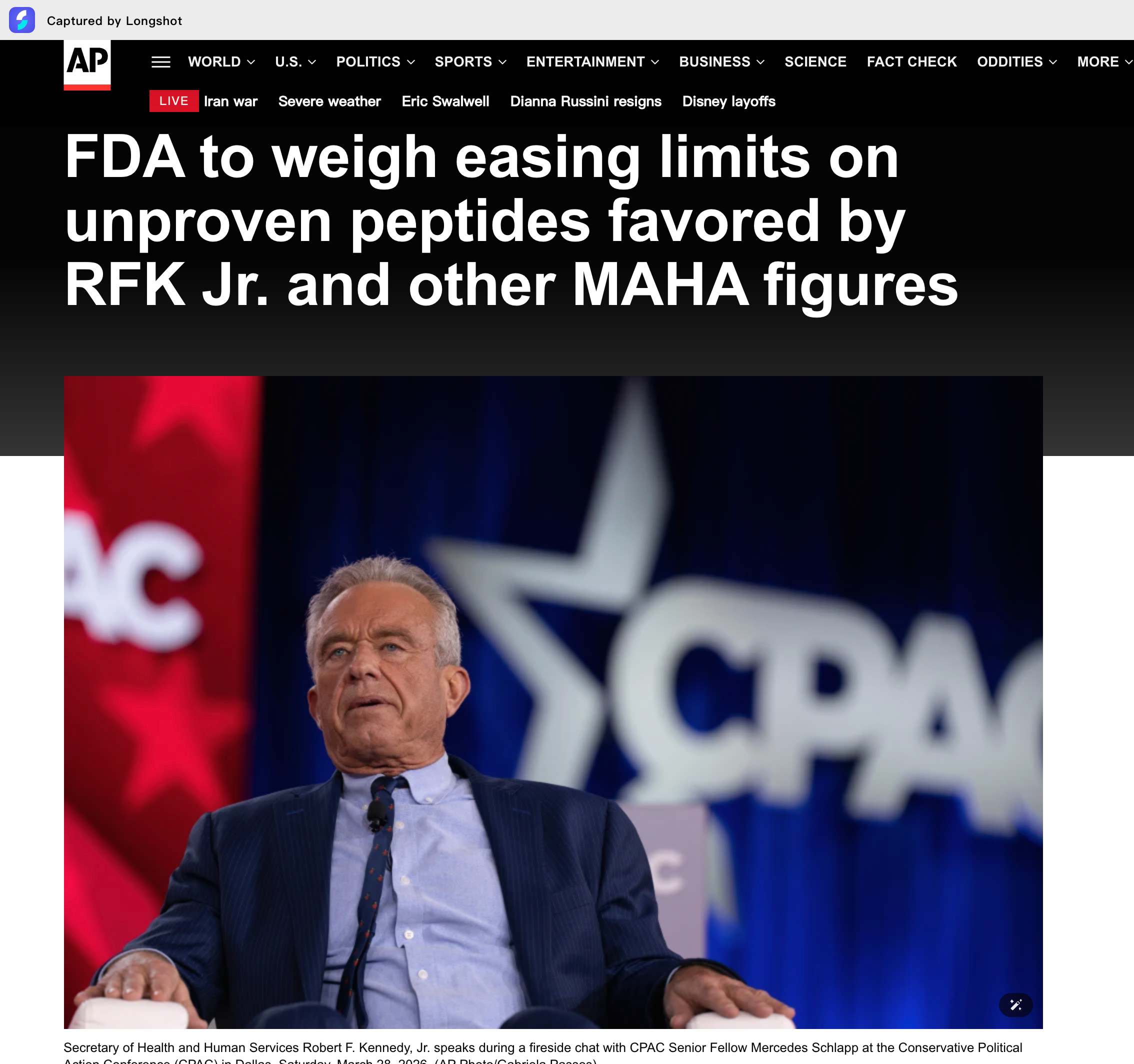
The meeting announcement on Wednesday came after Health Secretary Robert F. Kennedy Jr. repeatedly promised to ease regulations on peptide substances. Peptide substances are often promoted as a panacea for rapid muscle growth, wound healing, or anti-aging. However, these claims lack research support, and the safety of most peptide substances has not been reviewed by the U.S. Food and Drug Administration (FDA).
Kennedy has discussed using peptide drugs to treat his own injuries. Some of the main supporters of his "Make America Healthy Again" campaign are also active advocates of peptide drugs, including self-proclaimed "longevity expert" Gary Brecka, who sells various peptide formulations through his website.$Hims & Hers Health(HIMS.US) $Defiance Daily Target 2X Long HIMS ETF(HIMZ.US)
The copyright of this article belongs to the original author/organization.
The views expressed herein are solely those of the author and do not reflect the stance of the platform. The content is intended for investment reference purposes only and shall not be considered as investment advice. Please contact us if you have any questions or suggestions regarding the content services provided by the platform.